The following calculator can be used to assess what gauge pressure shall be set to reach the desired carbonation level, as well as how changes of temperature of an already saturated keg (e.g. at serving time) influence the saturation level and internal pressure, and how much gas is needed to both: force-carbonate and subsequently empty the keg.
The formulas used in the calculator assume that the kegged cider is initially saturated with CO2, which is the case when it is stored/aged under an airlock until the moment it is poured into the keg, and the pouring is done carefully in order not to loose much gas. Also, it assumes the empty volume in the keg is filled with pure CO2 (all air purged). Otherwise the target carbonation level will be lower than expected.
The model used is slightly simplified: it does not account for vapour pressures, changes of volume of the liquid, and possibly other details I did not even think about :). Also, it uses Henry’s law directly, without accounting for a slightly increased solubility of CO2 in a mix of water and ethanol.
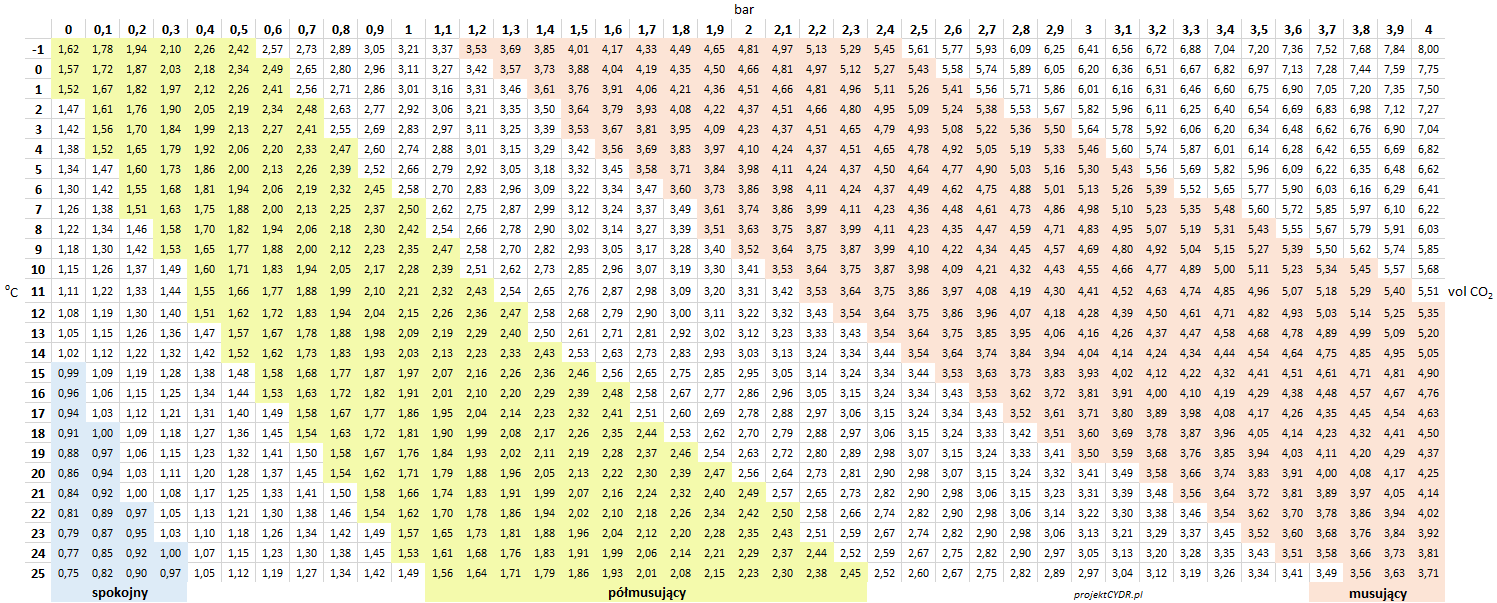
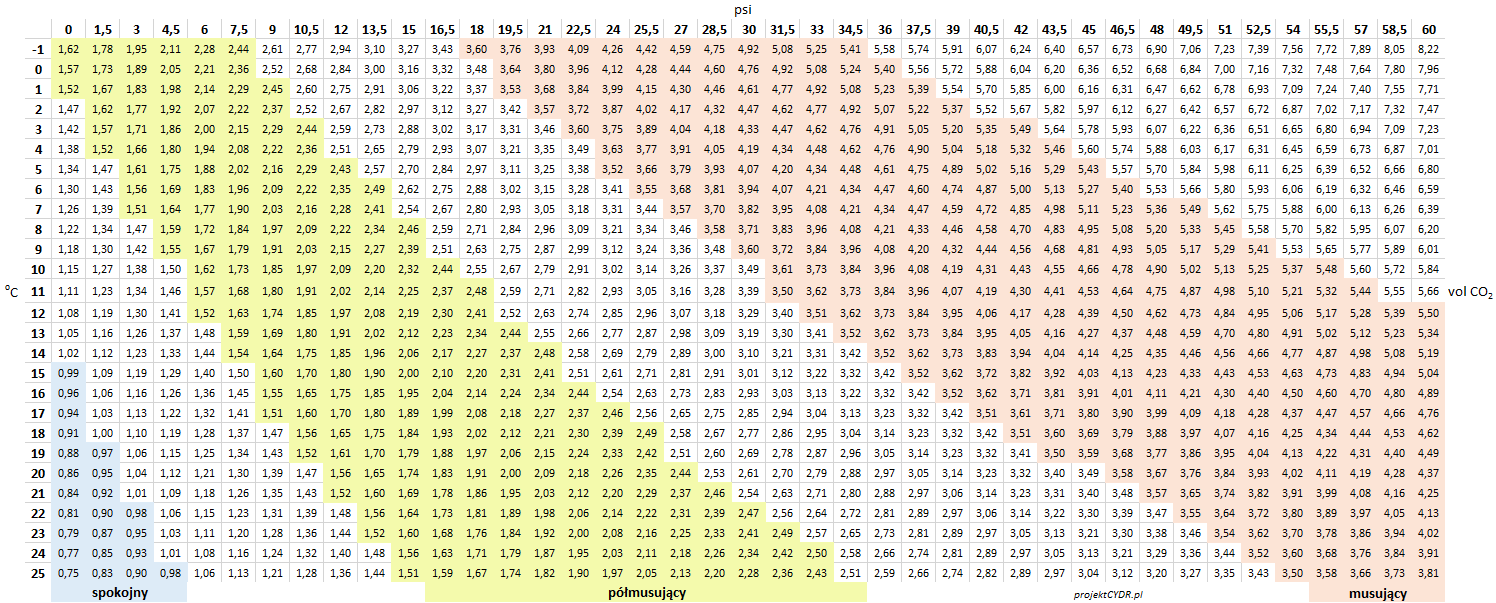
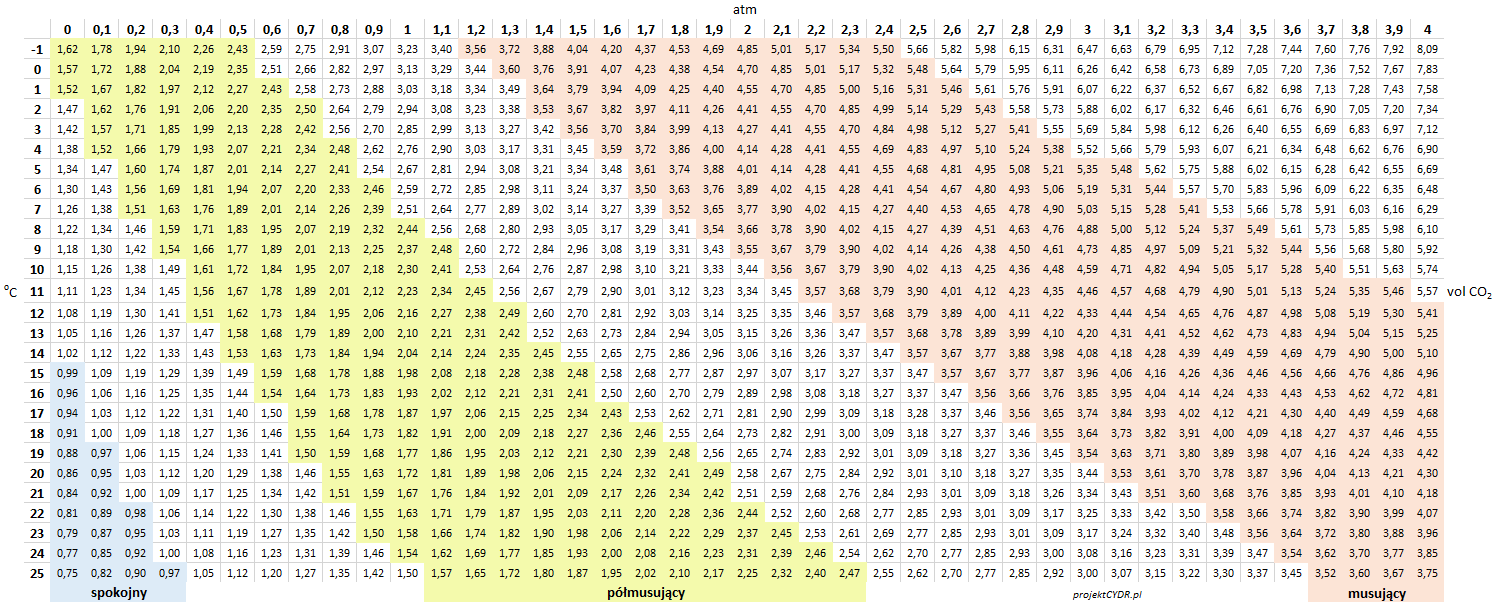
Tabulated data is available in the following tables.
pressure in psi:
pressure in atm:
For more background information ref. link (in Polish though..)